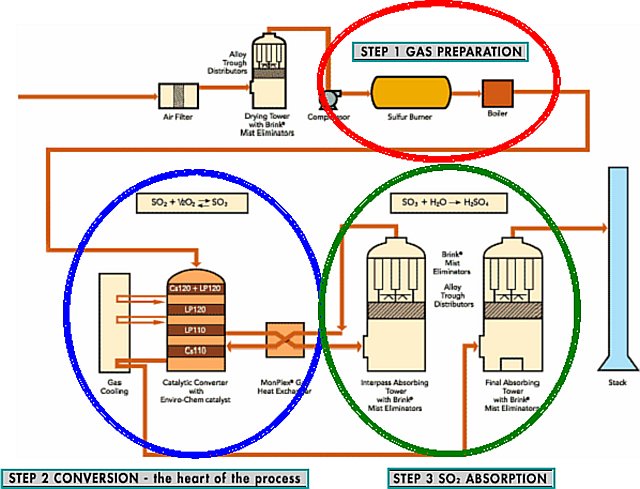
The Contact Process
1.Sulfur is burned to produce sulfur dioxide
Raw Materials
The first raw material is sulfur which is obtained from local refineries through being extracted and recovered from oil and gas. It's transported as a liquid and sampled and tested before being stored for production. The second raw material, Air is drawn from the atmosphere as dried and compressed.
Additional Information
Most of the worlds sulfuric acid is created and produced by the Contact Process. It's the most effective, efficient, and economically saving way up to date. Sulfuric acid is created in factories and plants. The main components that create sulfuric acid step by step are: sulfur furnace, converter, absorption towers, and the storage silo's. After the creation process is done, sulfuric acid is then sent off to consumers (mostly major companies and big buyers).
1.Sulfur is burned to produce sulfur dioxide
- S + O2 = SO2
- 2 SO2 + O2 ⇌ 2 SO3 (with V2O5
- SO3 + H2O = H2SO4
Raw Materials
The first raw material is sulfur which is obtained from local refineries through being extracted and recovered from oil and gas. It's transported as a liquid and sampled and tested before being stored for production. The second raw material, Air is drawn from the atmosphere as dried and compressed.
Additional Information
Most of the worlds sulfuric acid is created and produced by the Contact Process. It's the most effective, efficient, and economically saving way up to date. Sulfuric acid is created in factories and plants. The main components that create sulfuric acid step by step are: sulfur furnace, converter, absorption towers, and the storage silo's. After the creation process is done, sulfuric acid is then sent off to consumers (mostly major companies and big buyers).